Greg Tietjen, the CEO-Scientist-Musician Who Brings Organs Back to Life
Four funerals in four years strip life down to its essential frequencies. For Greg Tietjen, those years weren't just a season of grief; they were a masterclass in what happens when science loses its connection to the human soul. A Yale professor who thinks like a poet and plays guitar like a jazzman, Greg realized that the gap in medicine—the reason so many drugs fail—was afundamental disconnectbetween the laboratory and the human body, a bridge that had never been built. He decided to leave the safety of his tenured track to build that bridge himself, using the final, altruistic gifts of donors to create a new kind of 'Human Data.' This is the story of a man who decided that the best way to honor the people he lost was to change the way the world finds a cure.
The Jazz Guitarist Who Learned to Listen
Greg Tietjen's first degree was a Bachelor of Arts in English from Wake Forest University — he wanted to be a writer who could "translate the complexities of the human experience." But language, it turned out, wasn't his only instrument. Jazz guitar had been pulling at him since college, and after graduating, he followed it.
Then a soccer injury changed everything. After a shattered elbow and several surgeries, doctors told him he might never play guitar again.
Confined to a hospital bed, Tietjen felt what he would later describe as an "immense relief." He decided to turn the page and return to a childhood fascination with astrophysics, enrolling in physics at the University of Oregon. This conversion into fundamental science eventually led him to a PhD in biophysics at the University of Chicago and a faculty position at Yale.
From Laboratory to Operating Theater
In 2013, Tietjen arrived at Yale as a postdoctoral fellow in Biomedical Engineering. Four years later, he joined the faculty of the Yale School of Medicine in the Section of Transplantation and Immunology — a role that fused his engineering precision with clinical organ transplantation.
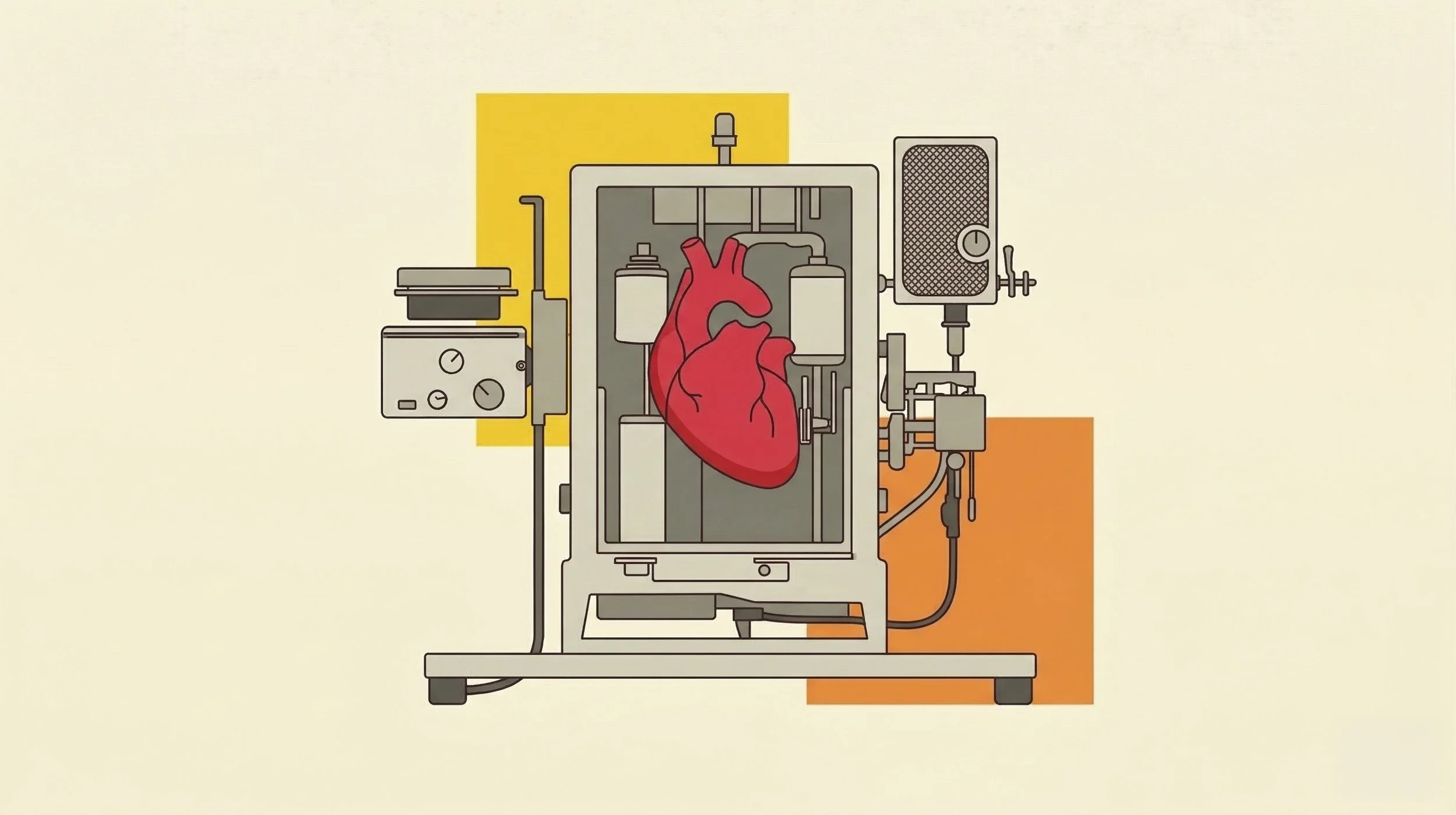
His laboratory became a pioneer in the emerging field of normothermic machine perfusion, a technology also being advanced by research groups at Cambridge and Oxford. But Tietjen saw an opportunity others missed: he chose to reimagine this clinical tool as a revolutionary platform for research.
Traditionally, organ preservation relied on static cold storage — essentially packing a human organ in ice and hoping it survived the journey. Normothermic perfusion, however, treats the organ as a living patient. By pumping oxygenated, nutrient-rich fluid through its veins at body temperature, the technology maintains the organ in a state of functional life outside the body. Tietjen’s team began calling them "mechanical patients" — a term that captured the shift from viewing an organ as a specimen to seeing it as a metabolizing, responding, and communicating entity.
The implications were staggering. A single donated organ could generate layer upon layer of molecular and physiological data—all from one human individual with a complete, de-identified medical history. No animal model could offer this fidelity; no cell culture could replicate this depth. This technology was the connective tissue between the petri dish and the patient, a missing link that medicine had been seeking for nearly a century.
In 1935, Alexis Carrel — the Nobel laureate who had mastered the vascular suture, the stitch that made organ transplantation possible — teamed up with the aviator Charles Lindbergh to build a glass pump that could keep an organ breathing outside the body. They documented their vision in a book titled The Culture of Organs. But where Carrel and his partner Lindbergh once struggled to keep organs pulsing in a lab, Tietjen saw a different opportunity. He didn't just want to keep the organs alive; he wanted to listen to what they were saying. He realized that these organs, while no longer viable for transplant, were the ultimate Human Data—a living laboratory capable of translating a promising molecule into a human cure.
Four Losses, One Calling
Then the losses came. Not in the abstract, clinical way that a transplant scientist encounters death daily, but in the intimate, devastating way that strips a life to its foundations.
"Revalia Biowas founded shortly after a four-year period where I lost my mom, my father-in-law, my aunt, and my uncle," Tietjen has said. "Each of them passed away under uniquely different and difficult circumstances. But each of their passings also left me with a profound new understanding of what matters most to me. I founded Revalia to honor their legacy by transforming how we innovate in biomedicine."
Viktor Frankl, the psychiatrist who survived Auschwitz, wrote that "in some ways suffering ceases to be suffering at the moment it finds a meaning." For Tietjen, meaning did not arrive gently. It arrived as an imperative. In March 2023, he walked away from a tenure-track position at Yale — one he was, by all accounts, close to securing — and co-founded Revalia Bio — a company built on the conviction that the dead could still help the living, if we had the tools to listen to what their organs had to say.
Rewriting the Rules of Drug Development
The average timeline for bringing a new therapy from laboratory to pharmacy stretches beyond a decade. The cost exceeds a billion dollars. And the primary reason for this: animal models do not adequately predict human biology.
The shift isn't about erasing animal models, but reordering them. It is about seeking human relevance first, then using animal studies as a supporting cast to map out the finer biological pathways where those models still excel.
"The old model of drug development is broken — decade-long timelines, 90% failure rates, and billion-dollar costs are no longer sustainable," Tietjen has stated. The regulatory landscape is shifting in his favor: recent changes at the FDA and NIH now actively seek to reduce dependence on animal testing, creating a window of opportunity that did not exist even five years ago.
Revalia's answer is what the company calls Human Data Trials — a new category of preclinical research that uses perfused, functional human organs to generate predictive data before any clinical trial begins. Their proprietary Human Data Stack integrates organ perfusion data with donor medical histories and digital analytics, creating a comprehensive picture of how a drug behaves in actual human tissue. Among the early milestones: a four-day kidney perfusion protocol and the development of the first human lung cancer model, created in collaboration with LifeShare of Oklahoma.
The very thing that makes human organ research messy — the enormous variability between donors — is precisely what makes it valuable. In tightly controlled animal studies, small treatment effects can achieve statistical significance but rarely survive contact with the diverse human population they are meant to serve. By embracing variability from the outset, Revalia's approach seeks to identify only those effects large enough to matter in the real world.
A New Paradigm in motion
The market response has been defined by striking velocity. Since its 2023 launch, Revalia has quadrupled its revenue and secured two of the world’s top ten pharmaceutical companies as customers. The pivot is led by industry veterans like Dr. Janet Nikolovski, who joined as Chief Data and Innovation Officer after 20 years at Johnson & Johnson. Her move signaled a broader institutional shift: major pharma had spent nearly a decade trying to internalize Human Data capabilities but lacked the specialized infrastructure and the stewardship model that Tietjen spent years perfecting at Yale.
"Drugs should be tested in the same biology, from the same patients, they are meant to treat."
The ethical dimension remains the company's core driver. As Chief Medical Officer Kourosh Saeb-Parsy notes:
"We see every donated organ as a legacy. Our job is to turn that gift into progress — not just for one trial, but for a new opportunity for developing life-saving medicines."
The Score No One Had Written
With $26.8 million in total funding — including a $14.5 million seed round co-led by America's Frontier Fund and Sierra Ventures, with the participation of Roger Ferguson, former Vice Chair of the U.S. Federal Reserve — and recognition as a 2026 TED Fellow, Tietjen has achieved in three years what many biotech founders spend a decade pursuing. Brian Wilcove of America's Frontier Fund called the ability to generate high-fidelity, human-specific data at scale "one of the most important advancements in biomedicine today."
This momentum was further validated in February 2026, when Revalia was awarded an ARPA-H contract worth up to $26.7 million to build the future of human-specific physiological modeling. It is a massive official endorsement of their work.
But Tietjen would probably say the numbers are not the point. What he has built is not merely a company. It is a proof of concept for a different kind of innovation — one that begins not with market analysis or technological capability, but with the irreducible human experience of loss. His trajectory — from jazz stage to English seminar to physics laboratory to surgical theater to startup — traces a path that no career counselor would ever recommend and no algorithm could predict.
In the quiet hum of Revalia’s perfusion machines, that journey finally makes sense. He hasn't just built a company; he has built a translator for what donors leave behind.
References:
Revalia Bio’s Quest to Replace Animal Models with Human Organs
Yale Technology Spotlight: Revalia Bio.
Albert, Claire and all. Honoring the gift: The transformative potential of transplant-declined human organs. American Journal of Transplantation 23 (2023) 165–170.